Virtual Laser Laboratory
About
|
Virtual laser laboratory (VLL) is a tool for 1D online simulation of laser experiments. Using VLL it is possible to run modeling, process results, draw plots, and download data files. Numerical method is based on a two-temperature hydrodynamic model. The model accounts for laser light absorption (solution to the electromagnetic wave equation), plume expansion (hydrodynamic motion), electron thermal conductivity, two-temperature effects (electron-phonon/ion collisions), phase transitions (melting, evaporation), material spallation (nucleation and growth). Among variety of possible problems that can be solved are pump-probe experiments with complex reflectivity measurements, ablation of multi-layer bulk and thin film targets, experiments on PHELIX-like set-ups, etc. It is possible to specify several pulses of different polarization, intensity profile, wavelength, right and left traveling direction. Basic metals such as Al, Cu, Au, Ag, Ni and others are available for target materials. There is a simple interface for preparation of task conditions and data plotting. Automatic or manual scale adjustment for axes and values is maintained while drawing. Basic Features
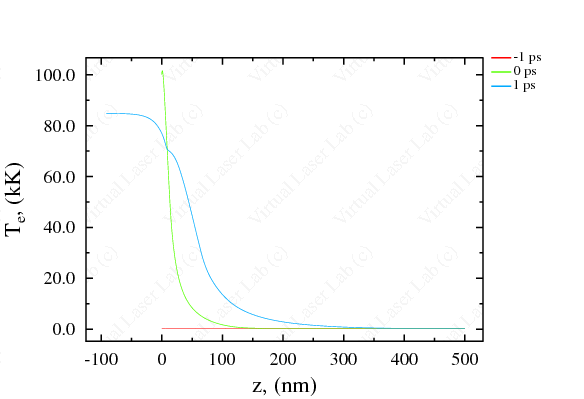
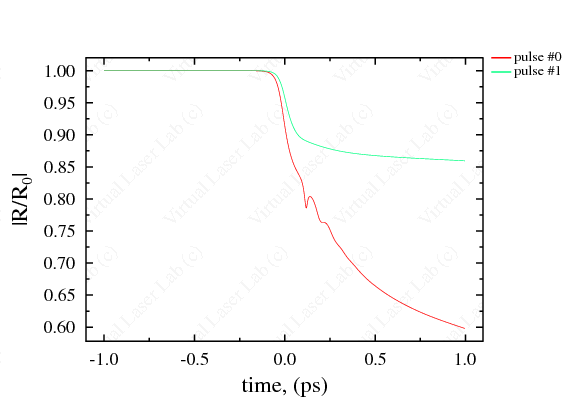
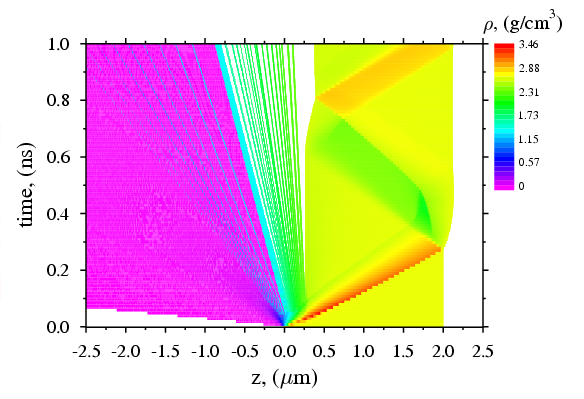
Available Parameters and Graphic Abilities Thermodynamic parameters: Space distribution of main hydrodynamic parameters such as density, temperature, pressure, velocity, mean charge of ions, and phase state in the target and plasma at the moments specified. Optic parameters: Time-dependent reflectivity, phase shift, transmission and absorption coefficients, including integral values. Time-space diagrams: Time-space 2D diagrams for density, pressure, temperature, phase state and mean charge of ions. |